Ionic EquilibriumHard
Question
Which is/are correct statements :
(a) When 100 ml of 0.1 M NaCN solution is titrated with 0.1 M HCl solution the variation of pH of solution with volume of HCl added will be (as shown in figure):
(b) Variation of degree of dissociation a with concentration for a weak electrolyte at a particular temperature is best represented by (as shown in figure) :
(c) 0.1 M acetic acid solution is titrated against 0.1 M NaOH solution. The difference in pH between 1/4 and 3/4 stages of neutralization of acid will be 2 log 3.
(a) When 100 ml of 0.1 M NaCN solution is titrated with 0.1 M HCl solution the variation of pH of solution with volume of HCl added will be (as shown in figure):
(b) Variation of degree of dissociation a with concentration for a weak electrolyte at a particular temperature is best represented by (as shown in figure) :
(c) 0.1 M acetic acid solution is titrated against 0.1 M NaOH solution. The difference in pH between 1/4 and 3/4 stages of neutralization of acid will be 2 log 3.
Options
A.a & c
B.b & c
C.a, b & c
D.b only
Solution
(a) Initially pH will decrease fast, then slowly due to buffer formation and then will decrease fast as buffer action diminishes.
(b) For a weak electrolyte
Ka =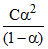
when α << 1 then α =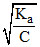
as C increases ⇒ αdecreases
as C is tending to zero ⇒ α will be unity
(c) At 1/4th neutralisation
CH3COOH + NaOH → CH3COONa + H2O
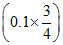
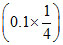
pH = pKa + log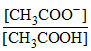 = pKa + log
= pKa + log 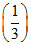
At 3/4th neutralisation
pH = pKa + log 3
so difference in pH = ᐃ(pH) = log 3 - log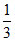 = 2 log 3
= 2 log 3
(b) For a weak electrolyte
Ka =
when α << 1 then α =
as C increases ⇒ αdecreases
as C is tending to zero ⇒ α will be unity
(c) At 1/4th neutralisation
CH3COOH + NaOH → CH3COONa + H2O
pH = pKa + log
At 3/4th neutralisation
pH = pKa + log 3
so difference in pH = ᐃ(pH) = log 3 - log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
0.2 millimoles of Zn2+ ion is mixed with (NH4)2 S of molarity 0.02 M. The amount of Zn2+ remains unprecipitated in 20 mL...In which of the following, the solubility of AgCl will be maximum?...Consider a titration of potassium dichromate solution with acidified Mohr′s salt solution using diphenylamine as i...A weak acid HA after treatment with 12 mL of 0.1 M strong base BOH has a pH of 5. At the end point the volume of same ba...The simultaneous solubilities of AgSCN and AgBr are, respectively (Ksp of AgSCN = 1 × 10–12, Ksp of AgBr = 2.1 × 10–13),...