Ionic EquilibriumHard
Question
Which of the following when mixed, will give a solution with pH > 7.
Options
A.0.1 M HCl + 0.2 M NaCl
B.100 mL of 0.2 M H2SO4 + 100 mL of 0.3 M NaOH
C.100 mL of 0.1 M HC2H3O2 + 100 mL of 0.1 M KOH
D.25 mL of 0.1 HNO3 + 25 mL of 0.1 M NH3
Solution
a. It is mixture of SA + salt of SA/SB. So pH is due to SA and less than 7.
b. mEq of H2SO4 = 100 × 0.2 × 2 = 40
mEq of NaOH = 100 × 0.3 = 30
So 30 mEq of NaOH will react with 30 mEq of H2SO4, So 10 mEq of H2SO4
(Strong acid will be left) So pH will be less than 7.
c. mEq of CH3COOH = 100 × 0.1 = 10
mEq of KOH = 100 × 0.1 = 10
So 10 mEq of WA(CH3COOH) will react with 10 mEq of KOH (SB) to give 10
mEq of salt (CH3COOH). It is a salt of WA/WB.
So its pH will be greater than 7.
pH =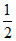 (pKw + pKa + logC)
(pKw + pKa + logC)
d. mEq of HNO3 = 25 × 0.1 = 2.5
mEq of NH3 = 25 × 0.1 = 2.5
So 2.5 mEq of HNO3 will react with 2.5 mEq of NH3 to give 2.5 mEq of NH4NO3,
Which is a salt of WB/SA, and its pH is always less than 7.
pH =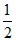 (pKw - pKb - log C)
(pKw - pKb - log C)
b. mEq of H2SO4 = 100 × 0.2 × 2 = 40
mEq of NaOH = 100 × 0.3 = 30
So 30 mEq of NaOH will react with 30 mEq of H2SO4, So 10 mEq of H2SO4
(Strong acid will be left) So pH will be less than 7.
c. mEq of CH3COOH = 100 × 0.1 = 10
mEq of KOH = 100 × 0.1 = 10
So 10 mEq of WA(CH3COOH) will react with 10 mEq of KOH (SB) to give 10
mEq of salt (CH3COOH). It is a salt of WA/WB.
So its pH will be greater than 7.
pH =
d. mEq of HNO3 = 25 × 0.1 = 2.5
mEq of NH3 = 25 × 0.1 = 2.5
So 2.5 mEq of HNO3 will react with 2.5 mEq of NH3 to give 2.5 mEq of NH4NO3,
Which is a salt of WB/SA, and its pH is always less than 7.
pH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A solution containing a mixture of 0.05 M NaCl and 0.05 M NaI is taken (Ksp of AgCl = 10-10 and Ksp of AgI = 4 × 10...Degree of hydrolysis for a salt of strong acid and weak base is...2.5 mL of M weak monoacidic base(Kb = 1 × 10-12 at 25o C) is titrated with M HCl in water at 25oC. The concentratio...Choose the correct statement...What mass of NaOH should be dissolved in sufficient water to get 20 m3 of an aqueous solution of pH 7.3 at 25o C?...