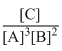Ionic EquilibriumHard
Question
The equilibrium constant for the following reaction will be: 3A + 2B → C
Options
A.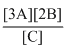
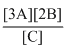
B.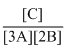
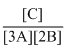
C.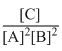
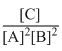
D.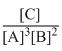
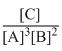
More Ionic Equilibrium Questions
100 ml of 0.5M hydrazoic acid (HN3, Ka = 3.6 × 10-4) and 400ml of 0.1M cyanic acid (HOCN, Ka = 8 × 10-4) are m...Which group of animals belong to the same phylum?...Among the following the weakest base is :-...Ka for HCN is 5 × 10-10 at 25oC. For maintaining a constant pH of 9, the volume of 5 M KCN solution required to be ...If the solubility of lithium sodium hexafluorido aluminate, Li3Na3 (AlF6)2 is ′s′ mol lt-1, its solubility p...