Ionic EquilibriumHard
Question
Ka for HCN is 5 × 10-10 at 25oC. For maintaining a constant pH of 9, the volume of 5 M KCN solution required to be added to 10 ml of 2M HCN solution is (log 2 = 0.3)
Options
A.4 ml
B.8 ml
C.2 ml
D.10 ml
Solution
Ka = 5 × 10-10 pKa = 10 log 5 = 9.3
pH = pKb + log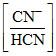
9 = 9.3 + log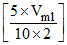 ⇒ - 0.3 = log
⇒ - 0.3 = log 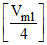
0.3 = log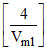 ⇒
⇒ 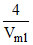 = 2 ⇒ Vml = 2 ml
= 2 ⇒ Vml = 2 ml
pH = pKb + log
9 = 9.3 + log
0.3 = log
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Solid Ba (NO3)2 is gradually dissolved in a 1.0 × 10-4 M Na2CO3 solution. At what concentration ofBa2+ will a preci...Which of the following compounds is not an antacid?...On adding 0.1 M solution each of [Ag+], [Ba2+], [Ca2+] in a Na2So4 solution, species first precipited is...Biodegradable polymer which can be produced fromglycine and aminocaproic acid is :-...During titration of acetic acid with aq. NaOH solution, the neutralisation graph has avertical line. This line indicates...