Ionic EquilibriumHard
Question
Among the following the weakest base is :-
Options
A.O2N-CH2-NH2
B.CH3-NH-CHO
C.C6H5-CH2-NH2
D.C6H5-CH2-NH-CH3
More Ionic Equilibrium Questions
An acid with molelcular formula C7H6O3 forms three types of sodium salts. i.e., C7H5O3Na, C7H4O3Na2 and C7H3O3Na3. The b...The dissociation constant of formic acid is 0.00024. The hydrogen ion concentration in 0.002 M-HCOOH solution is nearly...The pH of the solution obtanied by mixing 10 mL of 10-1 N HCI and 10 mL of 10-1 N NaOH is:...After solid SrCO3 was equilibrated with a buffer at pH 8.6, the solution was found to have [Sr2+] = 2.0 × 10−4 M, what i...The total number of different kind of buffers obtained during the titration of H3PO4 with NaOH are:...
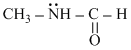 has delocalized l.p.
has delocalized l.p.