Ionic EquilibriumHard
Question
100 ml of 0.5M hydrazoic acid (HN3, Ka = 3.6 × 10-4) and 400ml of 0.1M cyanic acid (HOCN, Ka = 8 × 10-4) are mixed. which of the following is (are) true for the final solution?
Options
A.[H+] = 10-2 M
B.[N3-] = 3.6 × 10-3 M
C.[OCN-] = 6.4 × 10-3 M
D.[H+] = 1.4 × 10-2 M
Solution
Total [H+] = 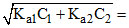
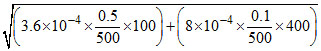
∴ [H+] = 10-2 M.
For HN3, [N3-] =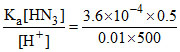 × 100 = 3.6 × 10-3 M.
× 100 = 3.6 × 10-3 M.
For HOCN, [OCN-] =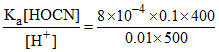 = 6.4 × 10-3 M.
= 6.4 × 10-3 M.
∴ [H+] = 10-2 M.
For HN3, [N3-] =
For HOCN, [OCN-] =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Calculate [S2–] in a solution originally having 0.1 M – HCl and 0.2 M – H2S. For H2S, Ka1 = 1.4 × 10−7 and Ka2 = 1.0 × 1...The reverse process of neutralisation is:...What is the pH of a neutral solution at 37°C, where Kw equals 2.5 × 10–14? (log 2 = 0.3)...If 50 ml of 0.2 M KOH is added to 40 ml of 0.5 M HCOOH. the pH of the resulting solution is: (Ka = 1.8 × 10-4, log ...Which of the following statements is correct for a solution saturated with AgCl and AgBr if their solubilities in moles ...