Chemical Kinetics and Nuclear ChemistryHard
Question
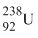 emits 8α - particles and 6β - particles. The netron /proton ratio in the product nucleus is :
emits 8α - particles and 6β - particles. The netron /proton ratio in the product nucleus is :Options
A.60/41
B.61/40
C.62/41
D.61/42
Solution
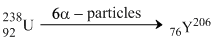

Neutrons = 206 - 82 = 124
Proton = 82
Ratio = 124 : 82 ⇒ 62 : 41
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Which is not true for a second order reaction ?...A first order reaction has a specific reaction rate of 10-2 sec–-1. How much time will it take for 20g of the reactant ...For the elementary reaction 2NO + O2 → 2NO2, the volume of a container is tripled. The rate becomes...For a first order reaction, (A) → products, theconcentration of A changes from 0.1 M to0.025 M in 40 minutes. The ...The rate law for a reaction between the substances A and B is given by rate = K[A]n [B]m. On doubling the concentration ...