Chemical Kinetics and Nuclear ChemistryHard
Question
Which is not true for a second order reaction ?
Options
A.It can have rate constant 1 × 10-2 L mol-1 s-1
B.Its half-life is inversely proportional to its initial concentration
C.Time to complete 75% reaction is twice of half-life
D.T50 = 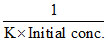
Solution
It is properties of Ist order Rxn and other all are properties of IInd order.
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
What is ᐃH for the reaction A + B → C, where the mechanism involves several kinetic steps....The bromination of acetone that occurs in acid solution is represented by CH3COCH3 (aq) + Br2 (aq) → CH3COCH2 Br (...A radioactive element has a half life of one day. After three days the amount of the element left will be :...Which reaction will occur at the fastest rate in water...The reaction A → B follows first order kinetics. The time taken for 0.8 mol of A to produce 0.6 mol of B is 1 hr. ...