NEET | 2017Chemical Kinetics and Nuclear ChemistryHard
Question
A first order reaction has a specific reaction rate of 10-2 sec–-1. How much time will it take for 20g of the reactant to reduce to 5 g ?
Options
A.
18.6 sec
B.
346.5 sec
C.
693.0 sec
D.
238.6 sec
Solution
Half life of first order reaction t1/2 =
Method-1
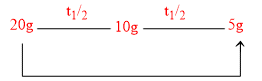
Total time = 2t1/2 = 2 × 69.3 = 138.6 sec
Method-2
t = 138.6 sec (Option 2)
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The rate of a first-order reaction is 0.04 mol l-1 s-1 at 10 second and 0.03 mol l-1 s-1 at 20 seconds afterinitiation o...The rate constant of first order reaction is 3 × 10-6 per second. the initial concentration is 0.10 M.The intial ra...t1/2 of a reaction: A → Products $\left( \text{Order = }\frac{3}{2} \right)$ is represented by $t_{1/2}\alpha\frac{1}{\l...3A → B + CIt would be a zero order reaction when :...A.G.M. counter is used to study the radioactive process of first-order. In absence of radioactive substance A, it counts...