SolutionHard
Question
The average osmotic pressure of human blood is 7.8 bar t 37oC. What is the concentration of an aqueous NaCl solution that could be used in the blood stream ?
Options
A.0.16 mol/L
B.0.31mol/L
C.0.60 mol/L
D.0.45 mol/L
Solution
π = cRT
7.8 = c × 0.082 × 310
c =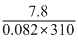 = 0.31 mol/L
= 0.31 mol/L
7.8 = c × 0.082 × 310
c =
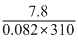 = 0.31 mol/L
= 0.31 mol/LCreate a free account to view solution
View Solution FreeMore Solution Questions
The vapour pressure of water is 12.3 kPa at 300 K. What is the vapour pressure of 1 molal aqueous solution of a non- vol...20 g of a substance were dissolved in 500 mL of water and the osmotic pressure of the solution was found to be 600 mm of...The diagram given below represents boiling point composition diagram of solution of component A and B, which is/are inco...Kf for water is 1.86 K kg mol-1. If your automobile radiator holds 1.0 kg of water, how many grams of ethylene glycol (C...At 80oC, the vapour pressure of pure liquid ′A′ is 520 mm Hg and that of pure liquid ′B′ is 1000...