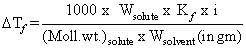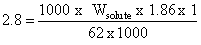SolutionHard
Question
Kf for water is 1.86 K kg mol-1. If your automobile radiator holds 1.0 kg of water, how many grams of ethylene glycol (C2H6O2) must you add to get the freezing point of the solution lowered to -2.8oC ?
Options
A.27 g
B.72 g
C.93 g
D.39 g
More Solution Questions
Due to osmosis, the volume of concentrated solutiona) decreases slowly (b) increases slowly (c) increases rapidly (d) de...The vapour pressure of pure benzene at 88°C is 960 mm and that of toluene at the same temperature is 380 mm of benzene. ...If 0.1 molal aqueous solution of sodium bromide freezes at −0.3348°C at one atmospheric pressure, then the percent disso...If α is the degree of dissociation of Na2SO4, the vant Hoff′s factor (i) used for calculating the molecular m...In a 0.2 molal aqueous solution of a weak acid HX the degree of ionization is 0.3. Taking kf for water as 1.85, the free...