SolutionHard
Question
The diagram given below represents boiling point composition diagram of solution of component A and B, which is/are incorrect among the following?
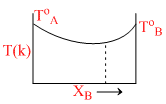

Options
A.The solution shows negative deviation
B.A-B-interactions are stronger than A-A and B-B
C.The solution is ideal solution
D.The solution shows positive deviation
Solution
For - ve deviation
A - - B > A - - A
A - - B > B - - B
- ve diviation solution are non ideal solution.
A - - B > A - - A
A - - B > B - - B
- ve diviation solution are non ideal solution.
Create a free account to view solution
View Solution FreeMore Solution Questions
Mole fraction of a non-electrolyte in aqueous solution is 0.07. If Kf is 1.86o mol-1 kg, depression in f.p., ᐃTf, ...Name the gas that can readily decolourise acidified KMnO4 solution :...The molar mass of substance forming 7.0% (by mass) solution in water which freezes at −0.93°C, is (The cryoscopic consta...The freezing point among the following equimolal aqueous solutions, will be highest for :...A vessel has nitrogen gas and water vapours in equilibrium with liquid water at a total pressure of 1 atm. The partial p...