SolutionHard
Question
20 g of a substance were dissolved in 500 mL of water and the osmotic pressure of the solution was found to be 600 mm of mercury at 15oC. The molucular weight of substance is :
Options
A.998
B.1028
C.1098
D.1198
Solution
πV = nRT
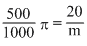 × 0.082 ×(273 + 15)
× 0.082 ×(273 + 15)
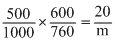 × 0.082 × 288
× 0.082 × 288
m =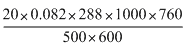 = 1198
= 1198
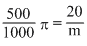 × 0.082 ×(273 + 15)
× 0.082 ×(273 + 15)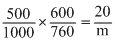 × 0.082 × 288
× 0.082 × 288m =
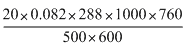 = 1198
= 1198Create a free account to view solution
View Solution FreeMore Solution Questions
H2S, a toxic gas with rotten egg like smell, is used for the quantitative analysis. If the solubility of H2S in water at...Aluminium phosphate is 100% ionised in 0.01 molal aqueous solution. Hence, ᐃTb / Kb is :...Osmotic pressure of 30% solution of glucose is 1.20 atm and that of 3.42% solution of cane sugar is 2.5 atm. The osmotic...The reagent that can distinguish between silver and lead salt is...Select correct statement ?...