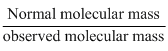SolutionHard
Question
Van′t Hoff factor is :
Options
A.More than one in case of association
B.less than one in case of dissociation
C.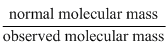
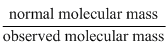
D.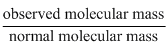
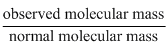
More Solution Questions
The molal boiling point elevation constant of water is 0.513°C kg mol−1. When 0.1 mole of sugar is dissolved in 200 g of...Aluminium phosphate is 100% ionised in 0.01 molal aqueous solution. Hence, ᐃTb / Kb is :...Electrolysis of dilute aqueous NaCl solution was carried out by passing 10 milli ampere current. The time required to li...An aqueous solution of a solute AB has b.p. of 101.08oC (AB is 100% ionised at boiling point of the solution) and freeze...Which of the following pairs of solution s are expected to be isotonic, temperature being the same ?...