SolutionHard
Question
Aluminium phosphate is 100% ionised in 0.01 molal aqueous solution. Hence, ᐃTb / Kb is :
Options
A.0.01
B.0.015
C.0.0175
D.0.02
Solution
AIPO4 ⇋ Al3+ + PO43-
i = 1 + x = 2
ᐃTb = molality Kbi
∴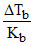 = 0.02.
= 0.02.
i = 1 + x = 2
ᐃTb = molality Kbi
∴
Create a free account to view solution
View Solution FreeMore Solution Questions
Osmosis involved in :...The product of hydrolysis of (P) and (Q) can be distinguished by -...Ratio of ᐃTb / Kb of 6% AB2 and 9% A2B (AB2 and A2B both are non-electrolytes) is 1 mol/kg in both cases. Hence, a...Thevan′t Hoff factor, i for a compound which undergoes dissociation in one solvent and association in other solven...When only a little quantity of HgCl2(s) is added to excess KI(aq) to obtain a clear solution, which of the following is ...