SolutionHard
Question
An aqueous solution of a solute AB has b.p. of 101.08oC (AB is 100% ionised at boiling point of the solution) and freezes at - 1.80oC. Hence, AB (Kb / Kf = 0.3)
Options
A.is 100% ionised at the f.p. of the solution
B.behaves as non-electrolyte at the f.p. of the solution
C.forms dimer
D.none of the above
Solution
Given ᐃTb = 1.08oC, i = 2 at boiling pt. of solution.
and ᐃTf = 1.80oC, and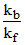 = 0.3]
= 0.3]
So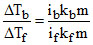
so if = 1
i.e., AB behaves as non–electrolyte at the f.p of the solution.
and ᐃTf = 1.80oC, and
So
so if = 1
i.e., AB behaves as non–electrolyte at the f.p of the solution.
Create a free account to view solution
View Solution FreeMore Solution Questions
Select correct statements :...PtCl4. 6H2O can exist as a hydrated complex 1 molal aq. solution has depression in freezing point of 3.72o. Assume 100% ...Two aqueous solutions S2 and S2 are separated by a semipermeable membrane. S1 has lower vapour pressure than S2. Which o...What is the concentration of Pb+2 when PbSO4 (Ksp = 1.8 × 10-8) begins to precipitate from a solution that is 0.004...Which facts are true when we use van′t Hoff equation PV = CST for osmotic pressure P of dilute solutions ?...