Mole ConceptHard
Question
60 gnm of an organic composed on analysis is found to have C = 24 g, H = 4 g and O = 32 g.The empirical formula of compound is :
Options
A.CH2O
B.CHO
C.C2H2O
D.C2H2O2
Solution
Ratio of no. of atoms = 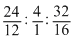 = 2 : 4 : 2 = 1 : 2 : 1
= 2 : 4 : 2 = 1 : 2 : 1
Empirical formula = CH2O.
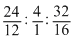 = 2 : 4 : 2 = 1 : 2 : 1
= 2 : 4 : 2 = 1 : 2 : 1Empirical formula = CH2O.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
For the reaction 2P + Q → R, 12 mol of P and 8 mol of Q are taken then...Consider the molecules CH4,NH3 and H2O. Which of the given statements is false?...If H2SO4 is formed from it′s elements by taking 6.023 × 1023 atom of ′O′ 5.6 litre of H2 gas and ...1.2575 g sample of [Cr(NH3)6]SO4Cl(Mw = 251.5) is dissolved to prepare 250mL solution showing and osmotic pressure of 1....The normality of solution obtained by mixing 10 ml of N/5 HCl and 30 ml of N/10HCl is :...