Mole ConceptHard
Question
If H2SO4 is formed from it′s elements by taking 6.023 × 1023 atom of ′O′ 5.6 litre of H2 gas and 8 gm S then
Options
A.0.125 moles of H2SO4 are formed
B.0.25 moles of H2SO4 are formed
C.no moles of ′S′ are left
D.1/4 mole of O2 is left
Solution
H2 + S + 2O2 → H2SO4
nH2 =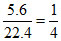 ns =
ns = 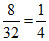 nO2 =
nO2 = 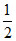
As all reactants are in stoichiometric ratios, none will be left behind.
Hence mole of H2SO4 is formed.
nH2 =
As all reactants are in stoichiometric ratios, none will be left behind.
Hence mole of H2SO4 is formed.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
One of N2 is mixed with 3 mole of H2 in a closed vessel of 3 ltr. 20% of N2 is converted into NH3 then Kc forN2 + H2 ...Nine volumes of gaseous mixture consisting of gaseous organic compound A and just sufficient amount of oxygen required f...Which of the following is dependent on temperature?...The density of 3M sodium thiosulphate is 1.25 g/ml. Identify the correct statements among the following :...Suppose you want an acidic solution to carry out a chemical reaction with 2 moles of NaOH. Which sample of acid is the b...