Mole ConceptHard
Question
1.2575 g sample of [Cr(NH3)6]SO4Cl(Mw = 251.5) is dissolved to prepare 250mL solution showing and osmotic pressure of 1.478 atm of Hg at 27oC. Which of the following statements is/are correct about this solution?
Options
A.Each molecule furnishes three ions in solution.
B.The Van′t Hoff factor is = 3.
C.The equilibrium molarity of [Cr(NH3)6]SO4Cl = 0.
D.The equilibrium molarity of [Cr(NH3)6]3+ = 0.02 M.
Solution
Molarity 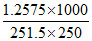 = 0.02 M, π = CRT
= 0.02 M, π = CRT
∴ πobs = 0.02 × 0.0821 × 300 = 0.4926 atm
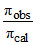 = i =
= i = 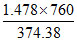 = 3
= 3
∵ α =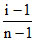 and = 1
and = 1
∴ n = 3
Also,
[Cr(NH3)6]SO4.Cl ⇋ [Cr(NH3)63+] + SO42- + Cl-
0.02 0 0 0
Eq conc 0 0.02 0.02 0.02
∴ πobs = 0.02 × 0.0821 × 300 = 0.4926 atm
∵ α =
∴ n = 3
Also,
[Cr(NH3)6]SO4.Cl ⇋ [Cr(NH3)63+] + SO42- + Cl-
0.02 0 0 0
Eq conc 0 0.02 0.02 0.02
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
In a compound C, H and N atoms are present in 9 : 1 : 35 by weight. Molecular weight of compound is 108. Molecular formu...The ratio of masses of oxygen and nitrogen in a particular gaseous mixture is 1 : 4. The ratio of number of their molecu...A solution containing Cu2+ and C2O42- ions which is titrated with 20 ml of M/4 KMnO4 solution in acidic medium completel...Cl - CH2 - CH2 - CH2 - Br + KCN → ? (1 mole) (1 mole)Product will be :-...The polymer in which the intermolecular force of attraction is weakest :-...