Mole ConceptHard
Question
For the reaction 2P + Q → R, 12 mol of P and 8 mol of Q are taken then
Options
A.3 mol of R is produced
B.6 mol of R is produced
C.25% of Q is left behind
D.25% of Q has reacted
Solution
2P + Q → R
initial mole 12 8 0
final mole 0 8 - 6 6
∴ moles of R formed = 6
% of Q left behind =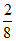 × 100 = 25%
× 100 = 25%
initial mole 12 8 0
final mole 0 8 - 6 6
∴ moles of R formed = 6
% of Q left behind =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Cl - CH2 - CH2 - CH2 - Br + KCN → ? (1 mole) (1 mole)Product will be :-...The weight of 1 L of ozonised oxygen at STP was found to be 1.5 g. When 100 mL of this mixture at STP was treated with t...Which of the following exihibit the weakest intermolecular forces :-...Some water is added to a mixture of 250 ml of 0.6 M HCl and 750 ml of 0.2 M HCl to obtain 0.25 M solution of HCl as a re...0.4 moles of NCl and 0.2 moles of CaCl2 were dissolved in water to have 50 mL of solution, the molarity of Cl ion is :...