NEET | 2016Mole ConceptHard
Question
Consider the molecules CH4,NH3 and H2O. Which of the given statements is false?
Options
A.
The H O H bond angle in CH4, the H N H bond angle in NH3, and the H O H bond angle in H2O are all greater than 90o .
B.
The H O H bond angle in H2O is larger than the H C H bond angle in CH4.
C.
The H O H bond angle in H2O is smaller than the H N H bond angle in NH3.
D.
The H O H bond angle in CH4 is larger than the H N H bond angle in NH3.
Solution
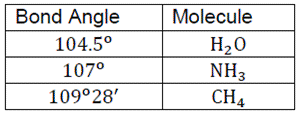
All the molecules are sp3 hybridized and Bond angle of H2O is smaller than NH3.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Law of multiple proportions is not applicable for the oxide(s) of...If 0.50 mole of BaCl2 is mixed with 0.20 mole of Na3 PO4, the maximum number of moles of Ba3(PO4)2 that can be formed is...Accumulation of which of the following molecules in the muscles occurs as a result of vigorous exercise ?...When a mixture of aluminium powder and iron (III) oxide is ignited, it produces molten iron and aluminium oxide. In an e...6.02 × 1020 molecules of urea are present in 100 ml of its solution. The concentration of urea solution is...