Atomic StructureHard
Question
The fossil bone has a 14C : 12C ratio, which is 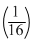 f that in a living animal bone. If the half life time of 14C is 5730 years, then age of the fossil bone is
f that in a living animal bone. If the half life time of 14C is 5730 years, then age of the fossil bone is
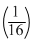 f that in a living animal bone. If the half life time of 14C is 5730 years, then age of the fossil bone is
f that in a living animal bone. If the half life time of 14C is 5730 years, then age of the fossil bone isOptions
A.11460years
B.17190years
C.22920years
D.45840years
Solution
Let 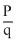 be the intial ratio of 14C : 12C. Let it is
be the intial ratio of 14C : 12C. Let it is 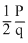 in the fossil. As q remains constant, p must have reduced to
in the fossil. As q remains constant, p must have reduced to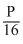 during the period.
during the period.
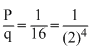
No. of half lives required = 4
Age of fossil = 4 × 5730 = 22920
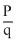 be the intial ratio of 14C : 12C. Let it is
be the intial ratio of 14C : 12C. Let it is 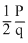 in the fossil. As q remains constant, p must have reduced to
in the fossil. As q remains constant, p must have reduced to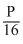 during the period.
during the period.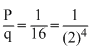
No. of half lives required = 4
Age of fossil = 4 × 5730 = 22920
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
If an electron and a photon propagate in the form of waves having the same wavelength, it implies that they have the sam...The wavelength of the first line of Lyman series for hydrogen atom is equal to that of thesecond line of Balmer series f...The transition from the state n = 4 to n = 3 in a hydrogen like atom results in ultraviolet radiation. Infrared radiatio...The activity of radioactive sample is measured as 9750 counts per minute at t = 0 and as 975 counts per minute at t = 5 ...Hydrogen (1H1), Deuterium (1H2) singly ionised Helium (2He4)+ and doubly ionised lithium (3Li6)++; all have one electron...