JEE Main | 2014Atomic StructureHard
Question
Hydrogen (1H1), Deuterium (1H2) singly ionised Helium (2He4)+ and doubly ionised lithium (3Li6)++; all have one electron around the nucleus. Consider an electron transition from n = 2 to n = 1. If the wave lengths of emitted radiation are λ1, λ2, λ3 and λ4 respectively then approximately which one of the following is correct?
Options
A.4λ1 = 2λ2 = 2λ3 = λ4
B.λ1 = 2λ2 = 2λ3 = λ4
C.λ1 = λ2 = 4λ3 = 9λ4
D.λ1 = 2λ2 = 3λ3 = 4λ4
Solution
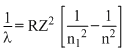
⇒ λ ∝
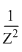 for given n1 & n2
for given n1 & n2⇒ λ1 = λ2 = 4λ3 = 9λ4
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Electrons with energy 80 keV are incident on the tungsten target of an X-ray tube. K-shell electrons of tungsten have a ...A full wave rectifier circuit along with the output is shown in figure. The contribution (s) from the diode 1 is (are)...A car is moving horizontally with a velocity v. A shell is fired upward with velocity u from the car. Find the horizonta...The Kα X-ray emission line of tungsten occurs at λ = 0.021 nm. The energy difference between K and L levels in...When cathode rays strike a metal target of high melting point with a very high velocity then which of the following are ...