Atomic StructureHard
Question
The wavelength of the first line of Lyman series for hydrogen atom is equal to that of thesecond line of Balmer series for a hydrogen like ion. The automic number Z of hydrogenlike ion is
Options
A.3
B.4
C.1
D.2
Solution
The wavelength of the first line of lyman series for hydrogen atom is
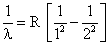
The wavelength of the second line of Balmer series for hydrogen line ion is
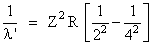
According to question λ = λ′
⇒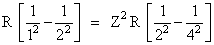
or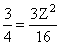 or Z2 = 4 or Z = 2
or Z2 = 4 or Z = 2
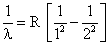
The wavelength of the second line of Balmer series for hydrogen line ion is
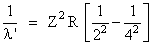
According to question λ = λ′
⇒
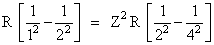
or
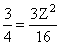 or Z2 = 4 or Z = 2
or Z2 = 4 or Z = 2Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Following are graphs of electric field variation with distance (r) for four different charge configurations.Choose the c...A proton is about 1840 times heavier than an electron when it is accelerated by a potential difference of 1 kV, its kine...A laser beam is used for carrying out surgery because it...A radioactive substance decays to 1/16th of its initial activity in to 40 days. The half -life of the radioactive substa...Let Mp be the mass of photon, mn the mass of neutron. M1 the mass of necleus and M2 the mass of nucleus. Then...