Atomic StructureHard
Question
If an electron and a photon propagate in the form of waves having the same wavelength, it implies that they have the same
Options
A.energy
B.momentum
C.velocity
D.angular momentum
Solution
A photon and electron will have same momentum if their wavelenght are equal.
For electron, momentum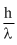
For photon, momentum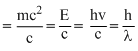
For electron, momentum
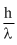
For photon, momentum
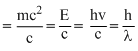
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The angular momentum of an electron in the 2nd excited state of a Helium ion (He+) is...When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the ...Consider the spectral line resulting from the transistion n = 2 → n = 1 in the atoms and ions given below. The sho...If the velocity of light were (2/3)rd of the present value, then the energy released in atomic explosion will be reduced...The transtion from the state n = 4 to n = 3 in a hydrogen like atom results in ultraviolet radiation. Infrared radiation...