Atomic StructureHard
Question
The transition from the state n = 4 to n = 3 in a hydrogen like atom results in ultraviolet radiation. Infrared radiation will be obtained in the transition from
Options
A.2 → 1
B.3 → 2
C.4 → 2
D.5 → 4
Solution
IR corresponds to least value of 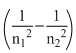
i.e. from Paschen, Bracket and Pfund series. Thus the transition corresponds to 5 → 4.
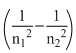
i.e. from Paschen, Bracket and Pfund series. Thus the transition corresponds to 5 → 4.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The wavelength of the first spectral line in the Balmer series of hydrogen atom is 6561 The wavelength of the second spe...Figure shows a system of three concentric metal shells A, B and C with radii a, 2a and 3a respectively. Shell B is earth...Assume that a neutron breaks into a proton andan electron. The energy released during thisprocess is : (Mass of neutron ...A shell is fired from a cannon with velocity n m/sec at an angle θ with the horizontal direction. At the highest po...The magnetic Resonance Imaging (MRI)is based on the phenomenon of...