Atomic StructureHard
Question
We wish to seen inside an atom. Assuming the atom to have a diameter of 10 pm, thismeans that one must be able to resolve a with of say 10 pm. if an electron microscope is used the minimum electron energy required is about
Options
A.1.5keV
B.15 keV
C.150keV
D.1.5MeV
Solution
From the de-Broglic equation,
λ =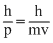
where I is wavelength, h is plank’s constant and p is momentum and v is velocity
v =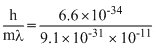 = 7.25 × 107 m/s
= 7.25 × 107 m/s
∴ Energy of electron =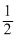 mv2
mv2
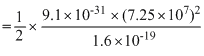 = 15 keV
= 15 keV
λ =
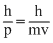
where I is wavelength, h is plank’s constant and p is momentum and v is velocity
v =
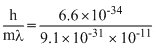 = 7.25 × 107 m/s
= 7.25 × 107 m/s∴ Energy of electron =
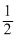 mv2
mv2 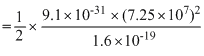 = 15 keV
= 15 keVCreate a free account to view solution
View Solution FreeMore Atomic Structure Questions
X-ray will not show the phenomenon of :...Let Mp be the mass of photon, mn the mass of neutron. M1 the mass of necleus and M2 the mass of nucleus. Then...For the same rise in temperature of one mole of gas at constant volume the heat required for non-linear triatomic gas is...The wavelength of the first spectral line in the Balmer series of hydrogen atom is 6561 The wavelength of the second spe...The energy of a photon of light with wavelength 5000 is approximately 2.5 eV. This way the energy of an X-ray photon wit...