Atomic StructureHard
Question
Let Mp be the mass of photon, mn the mass of neutron. M1 the mass of 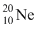 necleus and M2 the mass of
necleus and M2 the mass of 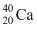 nucleus. Then
nucleus. Then
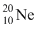 necleus and M2 the mass of
necleus and M2 the mass of 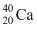 nucleus. Then
nucleus. Then Options
A.M2 = 2M1
B.M2 > 2M1
C.M2 < 2M1
D.M1 < 10(mp + nn)
Solution
Due to mass defect (which is finally responsible for the binding energy of the nucleus), mass of the nucleus is always less than the sum of masses of its constituent particles.
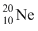 is made up 10 protons plus 10 neutrons. Therefore mass of
is made up 10 protons plus 10 neutrons. Therefore mass of 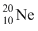 nucleus,
nucleus,
M1 < 10 (mp + mn)
Also, heavier the nucleus, more is the mass defect
Thus, 20 (mp + mn) - M2 > 10 (mp + mn) - M1
or 10(mp + mn) > M2 - M1
or M2 < M1 + 10 (mp + mn)
Now since, M1 < 10 (mp + mn)
∴ M2 < 2M1
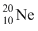 is made up 10 protons plus 10 neutrons. Therefore mass of
is made up 10 protons plus 10 neutrons. Therefore mass of 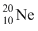 nucleus,
nucleus, M1 < 10 (mp + mn)
Also, heavier the nucleus, more is the mass defect
Thus, 20 (mp + mn) - M2 > 10 (mp + mn) - M1
or 10(mp + mn) > M2 - M1
or M2 < M1 + 10 (mp + mn)
Now since, M1 < 10 (mp + mn)
∴ M2 < 2M1
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Actinium 231, 231Ac89, emit in succession two β-particles, four alphas one β and one alpha plus serveral γ...the ratio of kinetic energy to the total energy of an electron in a Bohr orbit of the hydrogen atom, is :-...The wavelength of the first spectral line in the Balmer series of hydrogen atom is 6561 The wavelength of the second spe...When photo energy 4.25 eV strike the surface of a metal A, the ejected photoelectrons have maximum kinetic energy TA exp...The activity of radioactive sample is measured as 9750 counts per minute at t = 0 and as 975 counts per minute at t = 5 ...