Atomic StructureHard
Question
The energy of a photon of light with wavelength 5000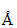 is approximately 2.5 eV. This way the energy of an X-ray photon with wavelngth 1
is approximately 2.5 eV. This way the energy of an X-ray photon with wavelngth 1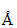 would be
would be
Options
A.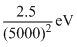
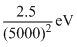
B.2.5 × 5000eV
C.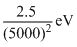
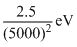
D.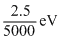
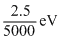
Solution
Energy of photon
E =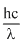
⇒ 2.5 eV =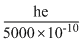 (asλ = 5000
(asλ = 5000  )
)
⇒ hc = 2.5 × 5 × 10-7eV
For X-ray photon,C =same,h = constant
⇒ E =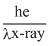
=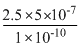 eV
eV
= 2.5 × 5000eV
E =
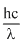
⇒ 2.5 eV =
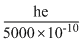 (asλ = 5000
(asλ = 5000 ⇒ hc = 2.5 × 5 × 10-7eV
For X-ray photon,C =same,h = constant
⇒ E =
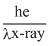
=
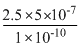 eV
eV= 2.5 × 5000eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Electric conduction in a semi-conductor takes place due to :-...The half life of a radioactive substance against α - decay is 1.2 × 107 s. What is the decay rate for 4.0 ...The function of heavy water in a nuclear reactor to...The maximum kinetic energy of photoelectrons emitted from a surface is $4 \text{ eV}$ when photons of energy $6 \text{ e...A mixture of 2 moles of helium gas (atomic mass = 4 amu) and 1 mole of argon gas (atomic mass = 40 amu) is kept at 300 K...