Atomic StructureHard
Question
For the same rise in temperature of one mole of gas at constant volume the heat required for non-linear triatomic gas is k times that required for monoatomic gas. What is the value of k:-
Options
A.0.5
B.1
C.1.5
D.2
Solution
Heat at constant volume for non linear triatomic gas = K (Heat for monoatomic gas)
μ(Cv)Tri dT = K(μCvdT)
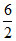 R = K
R = K 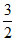 R
R
K = 2
μ(Cv)Tri dT = K(μCvdT)
K = 2
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Suppose the charge of a proton and an electron differ slightly. One of them is – e, the other is (e + ∆e). If the...The Kα X-ray emission line of tungsten occurs at λ = 0.021 nm. The energy difference between K and L levels in...Let Mp be the mass of photon, mn the mass of neutron. M1 the mass of necleus and M2 the mass of nucleus. Then...The kinetic in the potential different of 100V,...Photon of frequency v has a momentum associated with it. If c is the velocity of light, the momentum is...