Atomic StructureHard
Question
The wavelength of the first spectral line in the Balmer series of hydrogen atom is 6561 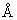 The wavelength of the second spectral line in the Balmer series of singly-ionized helium atom is
The wavelength of the second spectral line in the Balmer series of singly-ionized helium atom is
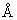 The wavelength of the second spectral line in the Balmer series of singly-ionized helium atom is
The wavelength of the second spectral line in the Balmer series of singly-ionized helium atom is Options
A.1215 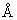
B.1640 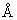
C.2430 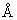
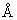
D.4687 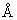
More Atomic Structure Questions
In photoelectric effected, the electrons are ejected from metals if the incident light has a certain minimum...The threshold wavelength for photoelectric emission from a material is $5200\ \text{Å}$. Photoelectrons will be emitted ...Selecte the correct statements from the following...Starting with a simple of pure 66Cu, of it decays in to Zn in 15 minutes. The corresponding half life is...Characteristic X-ray are produced due to...