Atomic StructureHard
Question
In Bohr′s theory. relation between principal quantum number n and radius of orbit r is :
Options
A.r ∝ 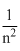
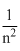
B.r ∝ n
C.r ∝ 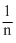
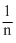
D.r ∝ n2
Solution
Radius of Bohr′s orbit ∝ n2, where n is prinsipal quantum no.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Consider an atom made up of a proton and a hypothetical particle that has double the mass of an electron but the same ch...Light of wavelength 4000 is incident on a metal plate whose work function is 2eV.What is maximum kinetic energy of emitt...If elements which principle quantum number n > 4 were not allowed in nature, the number of possible elements would be...One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure, The change ...In a hydrogen like atom in which every atom is in a particular excited state. Now a stream of photons of energy E0 bomba...