AIPMT | 2015Atomic StructureHard
Question
One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure,
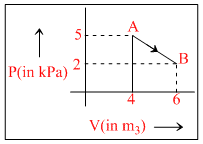
The change in internal energy of the gas during the transition is :

The change in internal energy of the gas during the transition is :
Options
A.-20 kJ
B.20 J
C.-12 kJ
D.20 kJ
Solution
ᐃU = nCvᐃT & T = 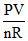
So ᐃT = T2 - T1 =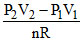
So ᐃU =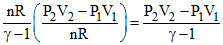
⇒ ᐃU =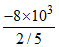 = - 20 kJ
= - 20 kJ
Hence option (1)
So ᐃT = T2 - T1 =
So ᐃU =
⇒ ᐃU =
Hence option (1)
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
If Alpha, Beta and Gamma rays same momentum, which has the longest wavelength...Nuclear reactions are given as -(i) ⎕ (n, p)15p32(ii) ⎕ (p, α)8O16(iii) 7N14 (⎕, p) 6C14 missing ...What is the exchange particle for strong nuclear interaction ?...A photon of 1.7 × 10-13 Joules is absorbed by a material under special circumstances. The correct statement is :-...When an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which in...