Atomic StructureHard
Question
Light of wavelength 4000 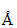 is incident on a metal plate whose work function is 2eV.What is maximum kinetic energy of emitted photoelectron?
is incident on a metal plate whose work function is 2eV.What is maximum kinetic energy of emitted photoelectron?
Options
A.0.5eV
B.1.1 eV
C.2.0 eV
D.1.5 eV
Solution
Applying Einstein′s equation
hv = φ +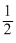 mv2 = φ + K.E
mv2 = φ + K.E
6.6 × 10-34 ×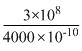
= 2 × 1.6 × 10-19 + K.E
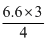 × 10-19 = 3.2 × 10-19 + K.E
× 10-19 = 3.2 × 10-19 + K.E
4.65 × 10-19 = 3.2 × 10-19 + K.E
K.E = (4.95 - 3.2) × 10-19
= 1.75 × 10-19 j.
= eV = 1.1eV
eV = 1.1eV
hv = φ +
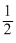 mv2 = φ + K.E
mv2 = φ + K.E6.6 × 10-34 ×
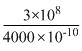
= 2 × 1.6 × 10-19 + K.E
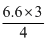 × 10-19 = 3.2 × 10-19 + K.E
× 10-19 = 3.2 × 10-19 + K.E4.65 × 10-19 = 3.2 × 10-19 + K.E
K.E = (4.95 - 3.2) × 10-19
= 1.75 × 10-19 j.
=
 eV = 1.1eV
eV = 1.1eVCreate a free account to view solution
View Solution FreeMore Atomic Structure Questions
A mixture of 2 moles of helium gas (atomic mass = 4 amu) and 1 mole of argon gas (atomic mass = 40 amu) is kept at 300 K...In Bohr′s theory. relation between principal quantum number n and radius of orbit r is :...What is the wavelength of the least energetic photon emitted in the Lyman series of the hydrogen spectrum ?...The minimum potential different between the base and emitter required to switch a silicon transistor ′ON′ is...Two identical p-n junctions may be connected in series with a battery in three ways. The potential drops across the two ...