Atomic StructureHard
Question
In a hydrogen like atom in which every atom is in a particular excited state. Now a stream of photons of energy 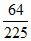 E0 bombarded into it and is absorbed by the hydrogen like atom and subsequently its emission spectrum shows 10 different lines in which some lines have energy less than
E0 bombarded into it and is absorbed by the hydrogen like atom and subsequently its emission spectrum shows 10 different lines in which some lines have energy less than 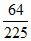 E0 and some lines have energy more than
E0 and some lines have energy more than 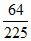 E0 and some lines have energy equal to
E0 and some lines have energy equal to 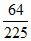 E0. (Where E0 is ionization energy of hydrogen atom in ground state).
E0. (Where E0 is ionization energy of hydrogen atom in ground state).
Options
A.The final quantum no. of electron in the atom is 5
B.The initial quantum no. of electron in the atom is 3
C.Atomic no. of the atom is 2
D.None of these
Solution
As emission spectrum of the atom shows 10 different lines, so the highest excited state of the electron is n = 5.
As some lines have energy less then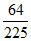 E0 and some have more than
E0 and some have more than 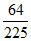 E0 and some have equal toit. So initial state of electron can be n = 2 or n = 3.
E0 and some have equal toit. So initial state of electron can be n = 2 or n = 3.
Again if initial level is n = 2, then Z2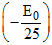 - Z2
- Z2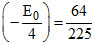 E0, Z is in fraction (not possible)
E0, Z is in fraction (not possible)
If n = 3, then
Z2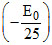 - Z2
- Z2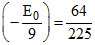 E0, Z = 2
E0, Z = 2
As some lines have energy less then
Again if initial level is n = 2, then Z2
If n = 3, then
Z2
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The ratio of the energy of the photon emitted the kα line to that of the kβ line is...If a source of power 4 kW produces 1020 photons/second, the radiation belong to a part of the spectrum called...Consider α- particle, β - particle and γ- particle each having an energy of 0.5 MeV. In increasing order ...Holes are charged carries in...The energy of a photon of light with wavelength 5000 is approximately 2.5 eV. This way the energy of an X-ray photon wit...