Ionic EquilibriumHard
Question
25 ml of a solution of barrium hydroxide on titration with a 0.1 molar solution of hydrochloric acid gave a litre value of 35 ml. The molarity of barium hydroxide solution was
Options
A.0.14
B.0.28
C.0.35
D.0.07
Solution
Ba(OH)2 + 2HCI → BaCl2 + 2H2O
Applying Molarity equation,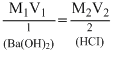 or 25 × M1 =
or 25 × M1 = 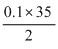 ∴ M1 =
∴ M1 = 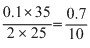 = 0.07
= 0.07
Applying Molarity equation,
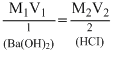 or 25 × M1 =
or 25 × M1 = 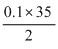 ∴ M1 =
∴ M1 = 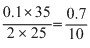 = 0.07
= 0.07Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The solubility of sparingly soluble salt A3B2 (molar mass = ‘M’ g/mol) in water is ‘x’ g/L. The ratio of molar concentra...The solubility product of BaCrO4 is 2.4 × 10-10 M2. The maximum concentration of Ba(NO3)2 possible without precipit...When equal volumes of the following solutions are mixed, precipitation of AgCl (Ksp = 1.8 × 10-1) will occur only w...The solubility of BaSO4 in water 2.42 × 103 gL–1 at 298 K. The value of solubility product (Ksp) will be (Gi...A saturated solution of silver benzoate (AgOCOC6H5) has pH of 8.6. Ka for benzoic acid is 5.0 × 10–5. The value of Ksp f...