Ionic EquilibriumHard
Question
When equal volumes of the following solutions are mixed, precipitation of AgCl
(Ksp = 1.8 × 10-1) will occur only with :
(Ksp = 1.8 × 10-1) will occur only with :
Options
A.10-4 M(Ag+) and 10-4 M(Cl-)
B.10-5 M(Ag+) and 10-5 M(Cl-)
C.10-6 M(Ag+) and 10-6 M(Cl-)
D.10-10 M(Ag+) and 10-10 M(Cl-)
Solution
For precipation
Ionic product > Ksp
Ionic product = [Ag+][Cl-]
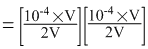
= 2.5 × 10-9 > Ksp
Thus precipitation will occur.
Ionic product > Ksp
Ionic product = [Ag+][Cl-]
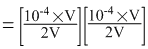
= 2.5 × 10-9 > Ksp
Thus precipitation will occur.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
At 30oC the solubility of Ag2CO3 (KSP = 8 × 10-12) would be greatest in one litre of:...Calculate the degree of hydrolysis of 0.005 M-K2CrO4. For H2CrO4, Ka1 = infinite, Ka2 =$5 \times 10^{- 7}$....Which would decrease the pH of 25 ml of a 0.01 M solution of hydrochloric acid? The addition of...If Ka1 and Ka2 of H2SO4 are 10-2 and 10-6 respectively then...What is the equilibrium constant of the following reaction? Fe(OH)3(s) + 3H3O+ $\rightleftharpoons$ Fe3+ + 6H2O?Ksp of ...