SolutionHard
Question
When during electrolysis of a solution of AgNO3 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposited on the cathode will be
Options
A.10.8 g
B.21.6 g
C.108 g
D.1.08 g
Solution
No. of moles of silver = 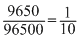 moles
moles
∴ Mass of silver deposited =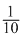 × 108 = 10.8g
× 108 = 10.8g
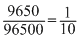 moles
moles∴ Mass of silver deposited =
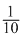 × 108 = 10.8g
× 108 = 10.8gCreate a free account to view solution
View Solution FreeMore Solution Questions
Two liquids X and Y form an ideal solution. At 300K, vapour pressure of the solution containing 1 mol of X and 3 mol of ...x mole of KCI and y mole of BaCl2 are both dissolved in 1 kg of water. Given that x + y = 0.1 and Kf for water is 1.85 K...Consider a binary mixture of volatile liquids. If at XA = 0.4 the vapour pressure of solution is 580 torr then the mixtu...When the depression in freezing point is carried out, the equilibrium exist between...A Solution of sucrose (molar mass = 342 gm-mol-1) has been prepared by dissolving 68.5 g of sucrose in 1000 g of water. ...