SolutionHard
Question
x mole of KCI and y mole of BaCl2 are both dissolved in 1 kg of water. Given that x + y = 0.1 and Kf for water is 1.85 K/molal, what is the observed range of ᐃTf, if the ratio of x to y is varied ?
Options
A.0.37o to 0.505o
B.0.185o to 0.93o
C.0.56o to 0.93o
D.0.37o to 0.93o
Solution
x + y = 0.1
x / y = 0.37
ᐃTf =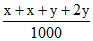 × 1000 × 1.85
× 1000 × 1.85
ᐃTf =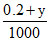 × 1000 × 1.85
× 1000 × 1.85
ᐃTf = (0.2 + 0.0729927) × 1.85 = 0.505
x / y = 0.37
ᐃTf =
ᐃTf =
ᐃTf = (0.2 + 0.0729927) × 1.85 = 0.505
Create a free account to view solution
View Solution FreeMore Solution Questions
An aqueous solution of hydrochloric acid...The molecular mass of NaCl as determined by osmotic pressure measurement is...There are some of the characteristics of the supersaturated solutionI : Equilibrium exists between solutions and solid s...An ionic compound that attracts atmospheric water so strongly that a hydrate is formed is said to be :...Total vapour pressure of mixture of 1 mol of volatile component A (pAo = 100 mmHg) and 3 mol of volatile component B (pB...