SolutionHard
Question
Two liquids X and Y form an ideal solution. At 300K, vapour pressure of the solution containing 1 mol of X and 3 mol of Y is 550 mm Hg. At the same temperature, if 1 mol of Y is further added to this solution, vapour pressure of the solution increases by 10 mm Hg. Vapour pressure (in mmHg) of X and Y in their pure states will be, respectively :
Options
A.200 and 300
B.300 and 400
C.400 and 600
D.500 and 600
Solution
PT = Pxoxx + Pyoxy
xx = mol fraction of X
xy = mol fraction of Y
∴ 550 = Pxo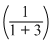 + Pyo
+ Pyo 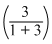
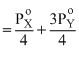
∴ 550 (4) = Pxo + 3Pyo ........(1)
Further 1 mol of Y is added and total pressure increases by 10 mm Hg.
∴ 550 + 10 = Pxo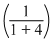 + Pyo
+ Pyo
∴ 560 (5) = Pxo + 4Pyo ........(2)
By solving (1) and (2)
We get, Pxo = 400 mm Hg
Pyo = 600 mm Hg
xx = mol fraction of X
xy = mol fraction of Y
∴ 550 = Pxo
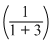 + Pyo
+ Pyo 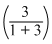
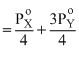
∴ 550 (4) = Pxo + 3Pyo ........(1)
Further 1 mol of Y is added and total pressure increases by 10 mm Hg.
∴ 550 + 10 = Pxo
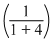 + Pyo
+ Pyo∴ 560 (5) = Pxo + 4Pyo ........(2)
By solving (1) and (2)
We get, Pxo = 400 mm Hg
Pyo = 600 mm Hg
Create a free account to view solution
View Solution FreeMore Solution Questions
A solution of ‘x’ mole of sucrose in 100 g of water freezes at − 0.2°C. As ice separates out, the freezing point goes do...Which of the following aqueous solution will have the highest boiling point?...Ice begins to separate at −0.744°C from an aqueous solution containing 2.4 g of a non-volatile, nonelectrolyte in 100 g ...Gold number of haemoglobin is 0.03. Hence, 1000 mL of gold sol will require haemoglobinso that gold is not coagulated by...Vapour pressure of CCl4 at 25oC is 143 mmHg. 0.5 g of a non-voltaile solute (molar mass = 65 mol-1) is dissolved in 100 ...