SolutionHard
Question
A Solution of sucrose (molar mass = 342 gm-mol-1) has been prepared by dissolving 68.5 g of sucrose in 1000 g of water. The Freezing point of the solution obtained will be
(Kf for water = 1.86 K kg mol-1)
(Kf for water = 1.86 K kg mol-1)
Options
A.- 0.372oC
B.- 0.520oC
C.+ 0.372oC
D.- 0.570oC
Solution
Depression in freezing point
ᐃ Tf = kf × m
where, m = molality =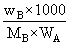
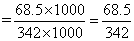
ᐃTf = 1.86 ×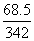
= 0.372oC
ᐃTf = To - Ts
= 0 - 0.372oC
= - 0.372oC
ᐃ Tf = kf × m
where, m = molality =
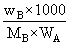
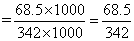
ᐃTf = 1.86 ×
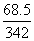
= 0.372oC
ᐃTf = To - Ts
= 0 - 0.372oC
= - 0.372oC
Create a free account to view solution
View Solution FreeMore Solution Questions
A liquid solvent is in equilibrium with its vapour. When a non-volatile solute is added to this liquid, the instant e ec...Consider following cases -I : 2M CH3COOH solution in benzene at 27oC where there is dimer formation to the extent of 100...The relationship between osmotic pressure at 273 K when 10 g glucose (P1) ,10 g urea (P2) and 10 g sucrose (P3) are diss...H2S on passing through ammonical solution gives white ppt. The white ppt. is of :-...72 g of glucose is dissolved in 1 kg of water in a sauce pan. At what temperature will the solution boil at 1.013 bar pr...