Solid StateHard
Question
Na and Mg crystallize in BCC and FCC type crystals respectively, then the number of atoms of Na and Mg present in the unit cell of their respective crystal is
Options
A.4 and 2
B.9 and 14
C.14 and 9
D.2 and 4
Solution
BCC - points are at corners and one in the centre of the unit cell
Number of atoms per unit cell = 8 ×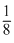 + 1 = 2
+ 1 = 2
FCC - points are at the corners and also centre of the six faces of each cell
Number of atoms per unit cell = 8 ×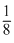 + 6 ×
+ 6 × 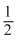 = 4
= 4
Number of atoms per unit cell = 8 ×
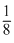 + 1 = 2
+ 1 = 2FCC - points are at the corners and also centre of the six faces of each cell
Number of atoms per unit cell = 8 ×
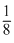 + 6 ×
+ 6 × 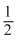 = 4
= 4Create a free account to view solution
View Solution FreeMore Solid State Questions
Consider a Body Centered Cubic(BCC) arrangement, let de, dfd, dbd be the distances between successive atoms located alon...The co-ordination number of fcc structure for metals is 12, since...The ionic radii of Cs and Cl are 0.165 nm and 0.181nm respectively. Their atomic weights are 133 and 35.5. Then,...NaCl is doped with 2 × 10–3 mole% SrCl2, the concentration of cation vacancies is (NA = 6 × 1023)...For the spinel structure (MgAl2O4), the correct statement is/are...