Solid StateHard
Question
Consider a Body Centered Cubic(BCC) arrangement, let de, dfd, dbd be the distances between successive atoms located along the edge, the face-diagonal, the body diagonal respectively in a unit cell.Their order is given by:
Options
A.de < dfd < dbd
B.dfd > dbd > de
C.dfd > de > dbd
D.dbd > de > dfd,
Solution
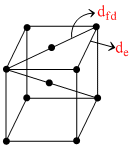
de = a
dfd = √2a
dbd =
∴ dfd > de > dbd
Create a free account to view solution
View Solution FreeMore Solid State Questions
A ferromagnetic substance becomes a permanent magnet when it is placed in a magnetic field because...The number of next nearest neighbours of $\text{Cs}^+$ ion in a $\text{CsCl}$ crystal is...The radius of Ag+ ion is 126 pm while of I− ion is 216 pm. The coordination number of Ag in AgI is...In a sodium chloride crystal, the unit cell edge length is a. The option(s) representing the correct combination is/areO...Calcium titanate crystallizes in a cubic unit cell with titanium atoms at the corner of the cell, oxygen atoms in the mi...