Solid StateHard
Question
The ionic radii of Cs and Cl are 0.165 nm and 0.181nm respectively. Their atomic weights are 133 and 35.5. Then,
Options
A.the lattice parameter (a) is 0.4 nm
B.the lattice parameter can not be determined from this data
C.the density of CsCI is 4.37 × 103 kg/m3
D.the CsCI structure has a fcc structure on the basis of given information.
Solution
For CsCl type, 2(r+ + r-) = a√3
So a =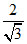 (0.165 + 0.181) = 0.4 mm
(0.165 + 0.181) = 0.4 mm
Density =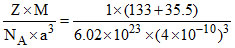 = 4.37 × 103 kg/m3.
= 4.37 × 103 kg/m3.
So a =
Density =
Create a free account to view solution
View Solution FreeMore Solid State Questions
A piece of copper and another of Ge are cooled from room temperature to 80 K. The resistance of:...An ionic solid is HCP of Q2− ions and Px+ ions are in half of the tetrahedral voids. The value of x should be...The composition of a sample of cuprous oxide is found to be Cu1.92O1.00 due to metal deficient defect. The molar ratio o...Which of the following statements are correct in context of point defects in a crystal ?...A compound of A and B crystallizes in a cubic lattice in which A atoms occupy the lattice points at the corners of a cub...