Solid StateHard
Question
The co-ordination number of fcc structure for metals is 12, since
Options
A.each atom touches 4 others in same layer, 3 in layer above and 3 in layer below.
B.each atom touches 4 others in same layer, 4 in layer above and 4 in layer below.
C.each atom touches 6 others in same layer, 3 in layer above and 3 in layer below.
D.each atom touches 3 others in same layer, 6 in layer above and 6 in layer below.
Solution
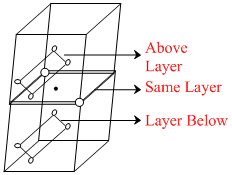
Fcc can be viewed in two following ways -
(i) Planes along the faces (and parallel to it) of the unit cell.
⇒ Each atom touches 4 in same layer, 4 in layer above and 4 in layer bellow it.
(ii) Planes along closest packed spheres → each atom touches 6 atom in same layer, 3 in layer above and 3 in layer below it.
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following is an intrinsic or thermodynamic defect?...Percentage of free space in cubic close packed structure and in body centred packed structure are respectively...Is there an expansion or contraction as iron transforms from FCC to BCC? The atomic radius of iron is 125 pm in FCC but ...An ionic solid of XY type having anions in CCP lattice and cations in the octahedral voids.Let a be the edge length of a...Which is the incorrect statement ?...