Solid StateHard
Question
For the spinel structure (MgAl2O4), the correct statement is/are
Options
A.50% OVs are occupied by ions.
B.Al3+ is equally distributed in TVs and OVs.
C.Oxide ions occupy ccp lattice.
D.12.5% TVs are occupied by ions.
Solution
For spinel structure, O2- ions form fcc arrangement.
∴ Number of O2- ions = 4,
Number of TV = 8, Number of OV = 4
Number of Mg2+ ions =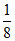 × TV =
× TV = 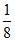 × 8 = 1
× 8 = 1
Number of Al3+ ions =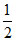 × OV =
× OV = 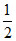 × 4 = 2
× 4 = 2
So, 50% OVs are occupied and 1/8th, i.e., 12.5% TVs are occupied,
∴ Number of O2- ions = 4,
Number of TV = 8, Number of OV = 4
Number of Mg2+ ions =
Number of Al3+ ions =
So, 50% OVs are occupied and 1/8th, i.e., 12.5% TVs are occupied,
Create a free account to view solution
View Solution FreeMore Solid State Questions
Antifluorite structure is derived from fluorite structure by :...The radius of Ag+ ion is 126 pm while of I− ion is 216 pm. The coordination number of Ag in AgI is...The ionic radii of Rb+ and I- are 1.46 and 2.16 (Ao). The most probable type of structure exhibited by it is...Which of the following statements is/are correct?...Cesium chloride on heating to 760 K changes into...