Atomic StructureHard
Question
If 13.6 eV energy is required to ionize the hydrogen atom, then the energy required to remove an electron from n = 2 is
Options
A.10.2 eV
B.0 eV
C.3.4 eV
D.6.8 eV
More Atomic Structure Questions
A hydrogen like atom of atomic number Z is in an excited state of quantum number 2n. It can emit a maximum energy photon...A gas is compressed isothermally to half its initial volume. The same gas is compressed separately throughan adiabatic p...A mixture of 2 moles of helium gas (atomic mass = 4 amu) and 1 mole of argon gas (atomic mass = 40 amu) is kept at 300 K...Out of the following which one is not a possible energy for a photon to be emitted by hydrogen atom according to Bohr...The transition from the state n = 3 to n = 1 in a hydrogen like atom results in ultraviolet radiation.Infrared radiation...
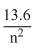 ⇒ E2 = -
⇒ E2 = - 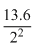 = 3.4eV
= 3.4eV