Atomic StructureHard
Question
A hydrogen like atom of atomic number Z is in an excited state of quantum number 2n. It can emit a maximum energy photon of 204 eV. If it makes a transition to quantum state n, a photon of energy 40.8 eV is emitted. The value of n will be
Options
A.1
B.2
C.3
D.4
Solution
Let ground state energy (in eV) be E1.
Then from the given condition
E2n - E1 = 204eV or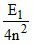 - E1 = 204eV
- E1 = 204eV
⇒ E1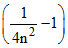 = 204 eV .....(i)
= 204 eV .....(i)
and E2n - En = 40.8 eV
⇒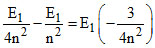 = 40.8 eV
= 40.8 eV
From equation (i) and (ii),
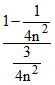 = 5 ⇒ n = 2
= 5 ⇒ n = 2
Then from the given condition
E2n - E1 = 204eV or
⇒ E1
and E2n - En = 40.8 eV
⇒
From equation (i) and (ii),
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In hydrogen atom spectrum, ( $R \rightarrow$ Rydberg's constant)A. the maximum wavelength of the radiation of Lyman seri...A stationary hydrogen atom of mass M emits a photon corresponding to the first line of lyman series. If R is the Rydberg...The energy of a photon of light with wavelength 5000 is approximately 2.5 eV. This way the energy of an X-ray photon wit...238U has 92 protons and 238 nucleons. It decays by emitting an Alpha particle and becomes...In an atom bomb, the energy is released because of the :...