Atomic StructureHard
Question
Out of the following which one is not a possible energy for a photon to be emitted by hydrogen atom according to Bohr′s atomic model?
Options
A.0.65 eV
B.1.9 eV
C.11.1 eV
D.13.6 eV
Solution
The energy of nth orbit of hydrogen atom is given as
En = -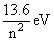
∴ E1 = - 13.6 eV
E2 = -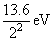 = - 3.4 eV
= - 3.4 eV
E3 = -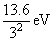 = - 1.5 eV
= - 1.5 eV
E4 = -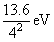 = - 0.85 eV
= - 0.85 eV
∴ E3 - E2 = - (1.5) - (- 3.4) = 1.9 eV
E4 - E3 = -(0.85) - (-1.5) = 0.65 eV
En = -
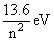
∴ E1 = - 13.6 eV
E2 = -
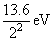 = - 3.4 eV
= - 3.4 eVE3 = -
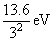 = - 1.5 eV
= - 1.5 eVE4 = -
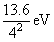 = - 0.85 eV
= - 0.85 eV∴ E3 - E2 = - (1.5) - (- 3.4) = 1.9 eV
E4 - E3 = -(0.85) - (-1.5) = 0.65 eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
What is the energy of He+ electron in first orbit ?...Consider α- particle, β - particle and γ- particle each having an energy of 0.5 MeV. In increasing order ...When an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which in...Characteristic X-ray are produced due to...An electron having charge e and mass m starts from lower plate of two metallic plates separated by a distance d. If pote...