NEET | 2016Atomic StructureHard
Question
A gas is compressed isothermally to half its initial volume. The same gas is compressed separately throughan adiabatic process until its volume is again reduced to half. Then:
Options
A.
Compressing the gas isothermally will require more work to be done.
B.
Compressing the gas through adiabatic process will require more work to be done.
C.
Compressing the gas isothermally or adiabatically will require the same amount of work.
D.
Which of the case (whether compression through isothermal or through adiabatic process) requiresmore work will depend upon the atomicity of the gas.
Solution
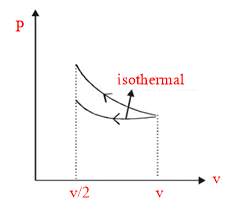
Isothermal curve lie below the adiabatic curve, So in adiabatic process more work to be done.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The electron in the hydrogen atom jumps from excited state (n = 3) to its ground state (n = 1) and the photons thus emit...Mark the INCORRECT statement :-...Which on of the following is a possible nuclear reaction...A radioactive material decays by simultaneous emission of two particles with respective half lives 1620 and 810 years. T...Two identical p-n junctions may be connected in series with a battery in three ways. The potential drops across the two ...